Why Manufacturing Controls Matter More Than Growing Conditions
In January, we laid out a core belief behind what we do at Blanco Creek Farms: Quality starts at the door. Inbound inspection is not a formality. It is a deliberate gate that protects customers by preventing out of specification raw produce from entering our system. That approach aligns with how regulators describe receiving and processing controls, including inspecting raw materials, storing them to prevent contamination and deterioration, and rejecting food that has become contaminated to the extent it is adulterated. [1]
This February post builds the next layer of the narrative: manufacturing rigor is the backbone of produce quality because manufacturing is the part of the supply chain where control is highest, outcomes are most measurable, and performance is most provable. Farms can reduce risk, but they operate in an open environment where variability is inherent and drivers can change quickly. FDA’s produce safety framework reflects this reality, including systems-based assessments for agricultural water that account for environmental and operational factors. [2]
By contrast, a processor can engineer repeatability through process design, documented SOPs, preventive controls programs, temperature control, sanitation systems, calibration and maintenance, training, verification sampling, statistical process control, and traceability-ready records. FDA’s preventive controls framework is built around precisely this kind of systematic, documented approach. [3]
For buyers in retail, food service, and procurement, this matters because a controlled manufacturing system reduces risk, improves spec consistency, stabilizes cost, and supports compliance and audit readiness. Traceability rules and GFSI-benchmarked audit programs further reinforce that the market expects documented, verifiable control, not just good intentions. [4]
The Control Map Where Quality is Actually Controllable
Quality in produce is the result of a chain, not a moment. The practical question is not whether growing conditions influence quality. They do. The buyer-focused question is: where can we most reliably reduce variability, detect problems early, and prove performance when it matters most.
A simple control map looks like this:
- Farm and harvest practices, including water and field conditions
- Transport and handling between harvest and receipt
- Receiving and inbound inspection
- Processing steps, including washing, cutting, packing, and cold chain control
- Storage, shipping, and distribution
- Customer receiving and handling
The key distinction is that the first half of this chain operates largely in open systems. The second half operates inside engineered systems. FDA’s produce safety regime includes standards for growing, harvesting, packing, and holding produce, which reflects the reality that risk exists upstream, but those risks are shaped by environmental and operational variables that can shift. [5]
When produce reaches a processor, expectations change. Current Good Manufacturing Practice requirements emphasize controlled conditions and controls to minimize contamination and deterioration. That includes receiving controls such as inspecting raw materials for cleanliness and suitability, storing to prevent contamination, and rejecting contaminated food. [1]
That is the thesis in operational terms: manufacturing is where quality becomes a system.
Growing Conditions Matter but They are Inherently Variable
The most important thing to say plainly is this: we respect our grower partners, and we recognize the skill it takes to produce consistent raw produce. But even the best growers operate under constraints that no one can fully command.
Weather, soil conditions, wildlife intrusion, and water quality are not static inputs. They are dynamic, and they can change faster than typical supply chain reaction time. Peer-reviewed research on preharvest transmission routes consistently describes multiple environmental niches and pathways that can introduce or spread pathogens in primary production ecosystems. [6]
Regulation aligns with that scientific reality. In late 2025, FDA finalized a preharvest agricultural water rule that replaced prior testing-based criteria with systems-based agricultural water assessments designed for hazard identification and risk management decision-making. Those assessments explicitly consider factors such as the nature of the water source, system protection from contamination, animal impacts, environmental conditions, and crop characteristics. [7]
This is not a critique of farming. It is the reason a processor must treat upstream variability as a given and design a downstream system that is resilient to it.
Growing Variability Versus Manufacturing Control
The table below summarizes how these two layers differ across dimensions buyers care about.
| Dimension | Growing-Level Variables | Manufacturing Controls |
| Controllability | Partial and distributed. Farms control practices, but key drivers such as weather, wildlife, and water system exposures can change. [8] | High and direct. Facilities can set conditions and controls for receiving, handling, sanitation, and storage, and can reject contaminated food. [9] |
| Predictability | Seasonal and event-driven. Risk can spike after environmental changes, and variability is inherent. [8] | Designed for repeatability. Preventive controls are built around monitoring, corrective actions, verification, and records. [3] |
| Measurability | Measurement is dispersed across acreage and time, and often indirect. [6] | Measurement is routine and record-based, including sanitation frequency, equipment condition, temperature controls, and supplier verification as applicable. [10] |
| Impact on Final Product | Sets the starting quality and risk profile but cannot guarantee uniform downstream outcomes. [11] | Strong influence on finished performance through controlled processing, cold chain stability, sanitation, and documented control of hazards. [12] |
Manufacturing Controls as the Backbone of Repeatable Quality
Manufacturing control is not one activity. It is an interlocking set of designed decisions that make performance repeatable. For a buyer, the most useful way to think about manufacturing rigor is that it creates three things: predictability, proof, and response speed.
The controls below are the backbone of that system.
Process Design and SOP Discipline
Facilities should be laid out to support controlled flow, prevent cross-contact, and reduce opportunities for contamination. CGMP expectations include conducting manufacturing under conditions and controls necessary to minimize the potential for microbial growth, contamination, and deterioration. [1]
SOPs are where that design becomes repeatable execution. A good SOP is not just a task list. It is a control document that defines who does what, when, to what standard, and what happens if the standard is not met. FDA’s preventive controls framework is built around documentation and execution, not ad hoc decision-making. [3]
HACCP Thinking and the PCQI Mindset
Buyers do not need a technical lecture on HACCP. They need to understand that a serious processor runs a hazard-based system focused on prevention.
FDA’s HACCP principles outline the logic of hazard analysis, identification of critical control points, critical limits, monitoring, corrective actions, verification, and record keeping. [13]
Under FSMA’s preventive controls approach, covered facilities develop and implement a food safety plan based on hazard analysis and risk-based preventive controls, supported by monitoring, corrective actions, verification, and records. [14]
The Preventive Controls Qualified Individual concept exists because preventive controls programs are only as strong as the competence behind them. Training and qualification expectations for individuals engaged in manufacturing include education, training, or experience, plus training in food hygiene and food safety principles appropriate to assigned duties. [15]
Temperature Control and Cold Chain Integrity
Cold chain control is one of the most practical examples of why manufacturing matters. A farm can harvest a good product. A processor can preserve that quality.
CGMP requirements emphasize holding food that can support rapid microbial growth at temperatures that prevent adulteration and conducting manufacturing under controls to minimize microbial growth and deterioration. [1]
Temperature control is also measurable, monitorable, and auditable. That matters to buyers who want to reduce shrink, avoid shelf-life surprises, and prevent product disputes.
Sanitation Systems and Environmental Control
Sanitation is not cleaning for cleanliness. It is a preventive control system for reducing contamination risk.
CGMP sanitary operations include cleaning and sanitizing food-contact surfaces as frequently as necessary to protect against contamination and, in wet processing, cleaning and sanitizing before use and after interruptions where surfaces may have become contaminated. [16]
This is especially important in produce processing environments where water can spread contamination if not controlled. CDC notes that if contaminated water or ice is used to wash, pack, or chill fruits or vegetables, contamination can spread to those items, and contaminated processing surfaces can spread germs to foods that touch them. [17]
For fresh-cut operations, FDA recommends environmental monitoring designed to detect pathogen harborage areas and verify effectiveness of cleaning and sanitizing programs, including sampling food-contact and non-food-contact surfaces. [18]
Calibration, Maintenance, and Equipment Condition
A buyer experiences equipment issues as inconsistent product. A processor manages that risk through maintenance, hygienic design, and calibration discipline.
CGMP requirements address equipment and utensil design, installation that facilitates cleaning and maintenance, and use that avoids adulteration. [19]
When instruments measure parameters tied to control, calibration matters because inaccurate measurements create false confidence. Preventive controls programs rely on monitoring and verification, which becomes fragile if measurement systems are not maintained. [20]
QA Sampling and Verification
Quality assurance cannot replace process control, but it can verify whether the system is working and detect drift early.
FDA’s preventive controls rule describes product testing and environmental monitoring as possible verification activities, required as appropriate to the food, facility, and role of the preventive control, and it notes that environmental monitoring is required in certain circumstances when contamination of ready-to-eat food with an environmental pathogen is a hazard requiring a preventive control. [21]
Statistical Process Control as an Early-Warning System
Statistical process control sounds technical, but the core idea is simple: use data trends to catch variation before it becomes a customer problem.
A systematic review of SPC implementation in the food industry describes it as a method used to assess and manage process variability, with reported motivations and benefits centered on improving performance and quality consistency. [22]
For produce processing, SPC can be applied to measurable attributes such as fill weights, defect rates, temperature stability, sanitizer concentration in wash water systems where used, or time-to-cool metrics.
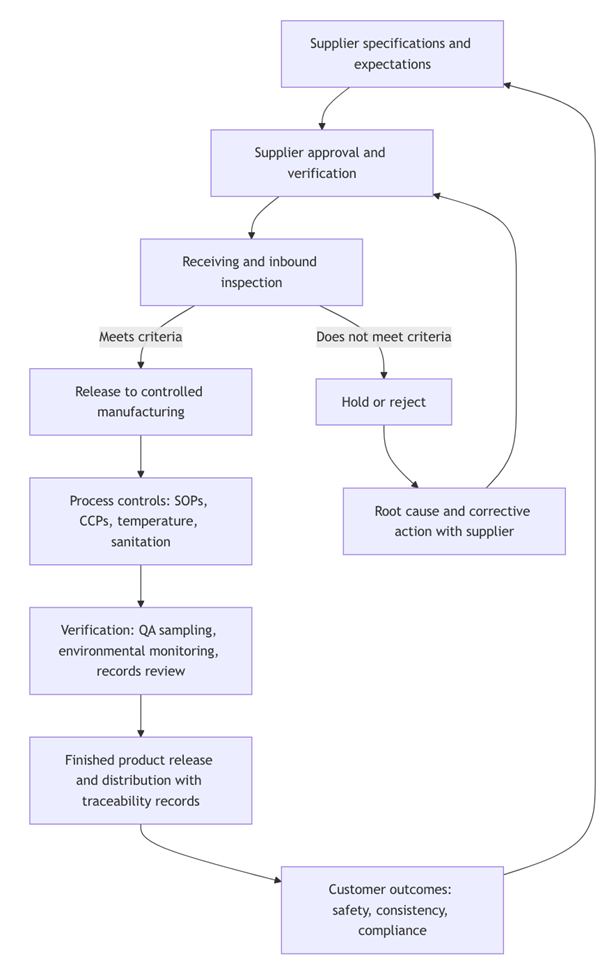
Connecting Inbound Inspection, Rejection, and Supplier Management
Manufacturing controls work best when the system begins at receiving.
Our January post emphasized the concept of a quality gate: inbound inspection protocols, rejection criteria, documentation, and the belief that saying no protects customers. That approach aligns with regulatory expectations that raw materials must be inspected and handled to ascertain they are clean and suitable, and that food that has become contaminated to the extent it is adulterated must be rejected or appropriately treated. [1]
Supplier Management as a Risk-Based Control
Sometimes, a hazard is best controlled upstream at the supplier level. FSMA’s preventive controls framework addresses this through a risk-based supply-chain program when hazard analysis identifies a hazard requiring a preventive control and the control will be applied in the supply chain. [21]
The supply-chain program concept is not abstract. The receiving facility must establish and implement a risk-based supply-chain program for certain raw materials and ingredients requiring supplier-applied controls. Appropriate supplier verification activities can include onsite audits, sampling and testing, and review of relevant food safety records. [23]
In practical terms, this is how inbound inspection integrates with supplier management:
- Clear, measurable inbound specifications
- Objective inspection and sampling at receipt
- Holds and rejections when criteria are not met
- Documented supplier corrective actions and trend-based performance review
- Tightened verification where risk or performance signals require it
How the System Protects Customers
What this Means for Buyers
Manufacturing rigor is not a “nice to have.” It directly affects buyer outcomes that show up on POs, contracts, scorecards, and profit-and-loss statements.
Risk Reduction and Compliance Confidence
Food safety risk is not only about whether a supplier cares. It is about whether they can demonstrate control through preventive systems and records. FDA’s preventive controls framework centers on written plans, monitoring, corrective actions, verification, and record keeping. [3]
Traceability expectations are rising as well. FDA’s Food Traceability rule requires parties who manufacture, process, pack, or hold foods on the Food Traceability List to maintain records with key data elements associated with critical tracking events and to provide information to FDA within 24 hours or within an agreed time frame. [24]
Consistency You Can Plan Around
Buyers care about consistent spec because inconsistency creates operational cost:
- Retail: Planogram performance, shrink, shelf-life complaints, brand trust
- Foodservice: Prep yields, portion consistency, labor planning, menu integrity
- Procurement: Predictability of cost and fewer disputes over spec compliance
USDA notes that quality grades provide a common language among buyers and sellers, supporting consistent quality for consumers. Manufacturing control is what translates that common language into repeatable product performance. [25]
Audit Readiness and Fewer Surprises
GFSI benchmarking is widely used as a confidence framework for food safety programs, and GFSI notes that recognized certification program owners meet benchmarking requirements that are widely accepted for food safety programs. [26]
For buyers, audit readiness is not about passing an audit. It is about whether the supplier’s daily operation is documented and disciplined enough that audits become a byproduct of good management.
Scenarios that Show Manufacturing Control Preventing Failures
The easiest way to see the difference between growing influence and manufacturing control is to imagine what happens when something goes wrong.
Scenario: Wash Water Turns a Small Problem into a Big Problem, or Stops It
If contaminated water or ice is used to wash produce, CDC notes contamination can spread to fruits and vegetables. [17] That means a processor’s wash system can either mitigate risk or amplify it.
Peer-reviewed research on produce wash water disinfectants highlights that cross-contamination potential in washing is highly dependent on water quality, and disinfection strategies are used to maintain water quality and reduce cross-contamination risk. [27]
A controlled manufacturing system responds by monitoring relevant parameters, maintaining sanitation standards, documenting results, and executing corrective actions when conditions move out of control. That is the difference between a contained deviation and a batch-wide event.
Scenario: Sanitation Drift Creates an Environmental Persistence Risk
FDA notes that Listeria monocytogenes can thrive in unsanitary food production conditions and can survive and grow under refrigeration. [28] CDC also describes how Listeria can spread in facilities through contaminated surfaces or equipment and that it can grow in refrigerated food. [29]
A manufacturing-focused quality system treats sanitation as a measurable control, not as a checklist. FDA’s fresh-cut guidance recommends environmental monitoring to detect pathogen harborage areas and verify sanitation effectiveness. [30]
A buyer experiences this as fewer recalls, fewer holds, fewer shelf-life surprises, and fewer brand-damaging outcomes.
Scenario: Inbound Variability Would Break the Line, but the Gate Prevents It
Imagine inbound raw product that visually looks acceptable at first glance but is outside specification in ways that threaten shelf life or processing yield. A processor that prioritizes throughput might push it through. A processor that prioritizes customers uses inbound inspection and clear disposition. CGMP expectations for inspecting raw materials and rejecting adulterated food exist because this gate is not optional if you want controlled outcomes. [1]
This is why the January post matters so much. The best manufacturing system in the world cannot compensate for raw materials that should never have entered it.
How to Measure and Build Manufacturing Control
Manufacturing rigor should be observable. Buyers should be able to ask questions and see evidence. Processors should be able to measure performance and improve it.
KPIs that Indicate Manufacturing Control is Working
The goal is not to drown your operation in metrics. The goal is to measure what signals control, drift, and responsiveness.
A practical KPI set includes:
- Inbound rejection rate and defect drivers by supplier and commodity, paired with corrective action closure rates, aligned to receiving and rejection expectations. [1]
- Preventive control monitoring completion rate and deviation frequency, aligned to preventive controls monitoring and verification expectations. [21]
- Temperature excursion rate for storage and staging, paired with time-to-correct, aligned to temperature control expectations. [1]
- Sanitation verification pass rate and environmental monitoring findings, aligned to sanitation and environmental monitoring guidance. [31]
- Calibration on-time rate and out-of-tolerance events for critical instruments, aligned to equipment and measurement reliability. [32]
- Training completion and retraining triggers tied to SOP changes, aligned to training and qualification expectations. [15]
- Traceability time-to-trace and record completeness for key tracking events, aligned to FDA traceability expectations. [33]
- Process capability measures for critical attributes using SPC where appropriate, aligned to documented benefits of SPC as variability management. [22]
Questions Buyers Should Ask Suppliers
Buyers often ask, “Are you certified?” That is a starting question. Stronger questions focus on control and evidence.
- What are your inbound acceptance criteria, and what percentage of loads do you reject or place on hold, with examples of documented disposition. [1]
- Do you operate under HACCP principles and a preventive controls food safety plan, and how do you define and monitor your control points. [34]
- How do you manage sanitation, including cleaning frequency, verification methods, and corrective actions when outcomes miss target. [16]
- Do you conduct environmental monitoring when appropriate, and how do you respond to findings to prevent recurrence. [35]
- How do you verify cold chain integrity from receiving through shipping, and can you provide recent records during supplier review. [1]
- How do you ensure instruments and controls are accurate, and what happens when a device is found out of tolerance. [32]
- If a hazard is controlled by your supply chain, what are your supplier verification activities and approval criteria. [23]
- How quickly can you provide traceability records if asked, and how do you manage key tracking events and data elements when applicable. [33]
An Implementation Roadmap for Processors
A credible roadmap focuses on layering repeatability, then proof, then speed.
Foundation and Quick wins. Start with disciplined receiving, documented acceptance criteria, and consistent product disposition rules. Reinforce sanitation frequency and tool control. Tighten temperature monitoring and staging practices. These are high-impact changes aligned with CGMP expectations and can be executed rapidly with training and oversight. [36]
System Build-Out. Formalize hazard analysis and preventive controls, clarify control points and corrective actions, and normalize records review as a routine management practice. Where environmental pathogens are relevant, build a fit-for-purpose environmental monitoring program and treat findings as a root-cause-driven improvement loop, not as an embarrassment. [37]
Supplier Integration. Where hazards are controlled by suppliers, implement a risk-based supply-chain program with defined verification activities, including audits, sampling and testing, and record review. Use inbound data to drive supplier scorecards, not just anecdotal feedback. [23]
Measurement Maturity. Introduce SPC for critical measurable attributes, starting small and scaling as the team builds confidence. A systematic review of SPC in food highlights that implementation is driven by variability management and performance improvement motivations, which is precisely what buyers experience as consistent product. [22]
Audit and Traceability Readiness. Ensure records are organized, consistent, and rapidly retrievable. For foods on the Food Traceability List, build systems that capture key data elements at required critical tracking events and support rapid response timelines. Align audit preparation to reality by ensuring the daily operation is the audit standard. [4]
Citations
[1] [9] [10] [12] [36] eCFR :: 21 CFR 117.80 — Processes and controls.
https://www.ecfr.gov/current/title-21/chapter-I/subchapter-B/part-117/subpart-B/section-117.80
[2] [7] [8] FSMA Final Rule on Pre-Harvest Agricultural Water | FDA
[3] [14] [20] [21] [37] FSMA Final Rule for Preventive Controls for Human Food | FDA
[4] [24] [33] FSMA Final Rule on Requirements for Additional Traceability Records for Certain Foods | FDA
[5] FSMA Final Rule on Produce Safety
[6] [11] Preharvest Transmission Routes of Fresh Produce Associated …
https://pmc.ncbi.nlm.nih.gov/articles/PMC6888529/?utm_source=chatgpt.com
[13] [34] HACCP Principles & Application Guidelines | FDA
[15] 21 CFR Ch. I (4–1–25 Edition) § 117.4
[16] [31] 21 CFR 117.35 — Sanitary operations.
[17] How Foodborne Outbreaks Happen
https://www.cdc.gov/foodborne-outbreaks/about/index.html?utm_source=chatgpt.com
[18] [30] [35] Guide on Microbial Hazards of Fresh-cut Fruits and …
[19] [32] 21 CFR 117.40 — Equipment and utensils.
[22] Review Statistical Process Control (SPC) in the food industry
https://www.sciencedirect.com/science/article/abs/pii/S0924224414000685?utm_source=chatgpt.com
[23] 21 CFR 117.405 — Requirement to establish and …
[25] Commodity Standards and Grades
[26] GFSI-Recognised Certification Programme Owners – MyGFSI
https://mygfsi.com/how-to-implement/recognition/certification-programme-owners/
[27] Effect of Disinfectants on Preventing the Cross-Contamination …
https://pmc.ncbi.nlm.nih.gov/articles/PMC4555240/?utm_source=chatgpt.com
[28] Listeria (Listeriosis)
https://www.fda.gov/food/foodborne-pathogens/listeria-listeriosis?utm_source=chatgpt.com
[29] How Listeria Spreads
https://www.cdc.gov/listeria/causes/index.html?utm_source=chatgpt.com
